
Taking your medical device prototype to ISO-13485 validated product launch
RD Support is a specialist company that bridges the gap between rapid prototyping and launching a mass-producible product.
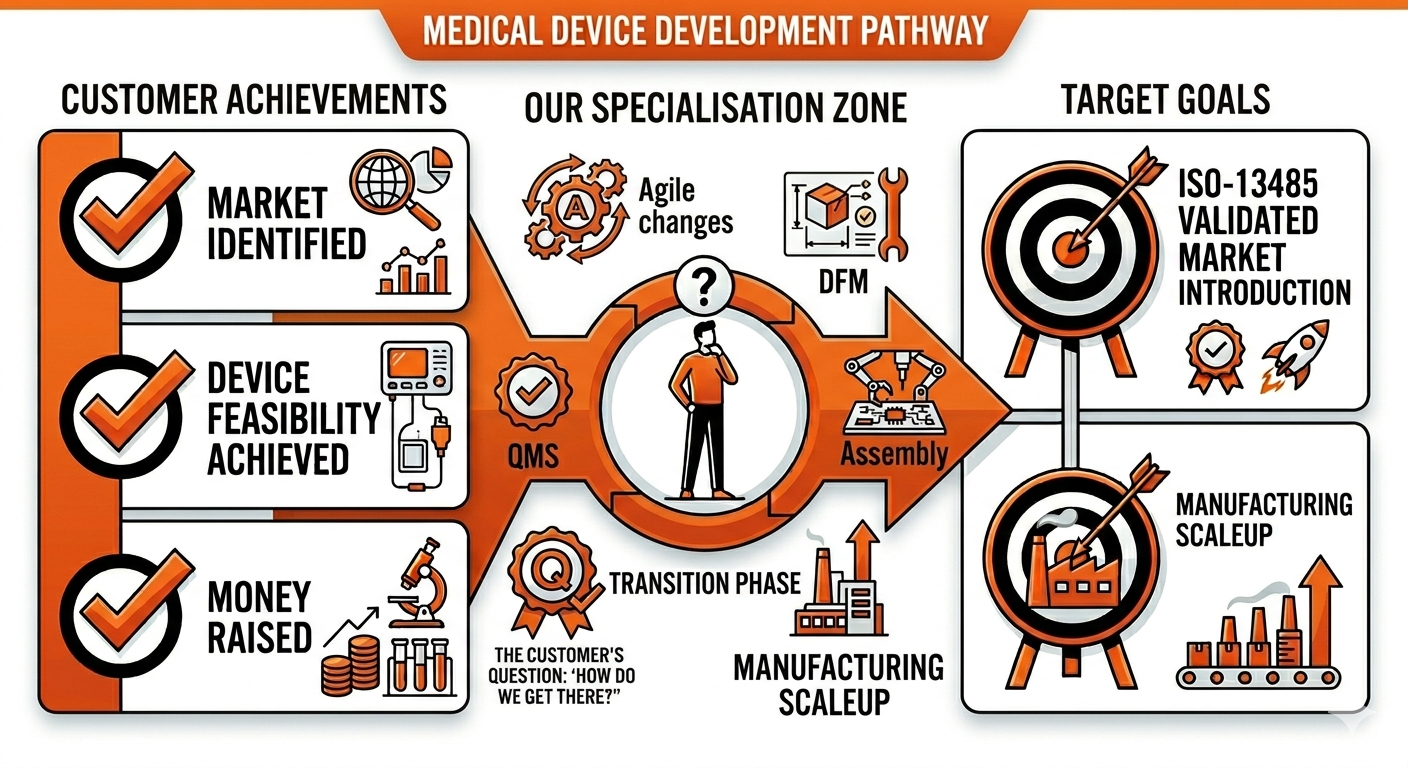
You have identified a new market opportunity for a medical device and have taken it all the way to a feasible device or lab protocol.
You are now mapping out your pathway to launch and are looking for someone to help you with design and manufacture.
You have been highly innovative, agile and efficient with your money and you’re not finding any global manufacturer to take your device to market.
This is because CMOs are great and cost-effective when the device design is frozen and validated and does not change anymore. They are not set up for this phase you are currently in. But we are!
RD-Support is specialised in taking medical products from Feasibility to validated product launch.
RD Support is expert at getting your product to market.


Our Services

RD Support bridges the gap between innovative diagnostic development and mass production, offering a unique fast and flexible manufacturing model. Unlike traditional CMOs focused on high-volume, frozen designs, or legal manufacturers constrained by regulatory requirements, RD Support specializes in rapid iteration with daily changes while maintaining focus on ISO13485 certification.
With extensive in-house IVD and DFM expertise, production-quality toolmaking, and full cleanroom facilities, we enable seamless transition from development through clinical trials to commercial launch.
We provide the agility of a development partner you want with the quality standards of regulated manufacturing you need.
From medical device prototype to certified product on the market in 3 simple steps:

1 Design For Manufacture
We take your proven lab protocol and/or design inputs (the product specification) and run it through our proven DFM (design for manufacture) process to ensure that every component and every assembly step is scalable and cost effective in volume manufacturing.
Elements can be rapidly proven by rapid prototyping.

2 Tooling
We’ll manufacture production-quality mould tools that we can modify every day if required. We update our cleanroom assembly equipment and adjust our processes to meet your throughput and quality expectations at every phase of the project.
We send you development batches and make agile changes until the performance is right.

3 Manufacturing
We validate our component manufacture and assembly processes.
We produce verification and validation batches and documentation.
We deliver PQ (product quality) batches to support your regulatory approval and market supply.
4 High Volume Production
Once on the market and production quantity ramps up significantly, we’ll provide you with a competitive piece price, or we’ll transfer manufacture to your CMO of choice, if desired.






